|
12/8/2023 0 Comments Carbon 14 atomic number
So we look at the mass number here, so 14 minus seven gives us seven neutrons. Seven protons, and 14 minusħ gives us seven neutrons. Our atomic number is seven, so we have seven protons, let's go ahead and write that here. The carbon-14 nucleus is turning into the nucleus for nitrogen here. An electron has a negative one charge, let's write a negative oneĬharge here for the electron. Here for carbon, we have six protons, let me go and write Has a negative one charge, and so that's what we're It's a conversion that's governedīy the weak nuclear force. The carbon-14 nucleus is actually going to give off an electron, and so that's pretty weird, and we'll talk about more And when you're writing nuclear equations, you're representing only the nuclei here, so for example, on the left side of my nuclear equation, I have carbon-14, we're talking about only the nucleus, so we're talking about six protons and eight neutrons in the nucleus. So let's look at the nuclear equation which represents the It's going to try to get a better neutron to proton ratio. The carbon-14 nucleus is unstable, it's radioactive, it's going to undergo spontaneous decay. Is greater than one, so we have an unstable nucleus. So what's the neutron to proton ratio? So what's the N to Z ratio here? Well the N to Z ratio would be eight neutrons and six protons, and obviously that number Number of neutrons will be 14 minus six, so eight neutrons. We have carbon-14, so let's get a little space right down here. So when N over Z is equal to one, you can say you have a stable nucleus, so equal numbers of protons and neutrons turns out to be stable. That have small numbers of protons, so if we're talkingĪbout Z is less than 20, they have stable nuclei when the N to Z ratio is equal to one. So if we're concerned with the ratio, the ratio of neutrons to protons, so the N to Z ratio. 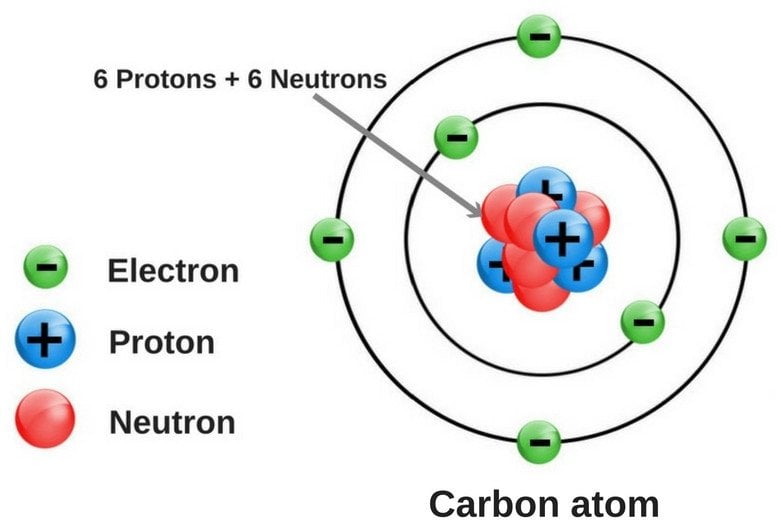 
So let's think about the atomic number, which tells us the number of protons, which we represent by Z. Of protons and neutrons, and so that's interesting. The point is that this nucleus is stable. That's not really the point of this video. You can get into much more detail about the strong force. Proton-neutron interaction, which is the same as a For example, a proton-proton interaction is the same as a The strong force acts only over short distances though, but it does act between all nucleons. So the nuclear strong force is stronger than the electrostatic force. We know that this nucleus is stable, so there must be something else holding the nucleus together, which we call the strong force. And so these two protons are repelling each other, and that's Here's a picture of the nucleus, with two protons and two neutrons, and we know it's stable, even though we know like charges repel. Protons and neutrons in the nucleus are called nucleons, and so I'll use that term a few times in this video. Video, we talked about the helium nucleus, which contains two protons and two neutrons. This is a simplification but it gives you the idea that you cannot think of each particle in isolation. Better to be a repelled proton than an unstable neutron, basically. 
The overall arrangement becomes more stable once one of those unstable almost-free neutrons becomes a nice, stable proton, even though the proton is repelled by other protons in the nucleus. Something has to give to enable the overall state of the nucleus to become more stable. But remember that free neutrons decay into protons? Well this guy on the outside is not quite free but he's not in a position to be stable. You can sort of imagine there is a neutron on the outside of the nucleus, and if it could get in and be surrounded by more particles, maybe it would be stable. Consider how C-14 decays: one of the neutrons turns into a proton. When you add one more to the pile, even if it will "stick" because of strong nuclear force, it changes the overall arrangement of all the protons and neutrons, and some arrangements are just more stable than others. You have all these particles trying to find a stable arrangement. When you add a neutron to C-13 to get C-14, yes there is more strong nuclear force but you have to consider the nucleus as a whole. They will decay into protons, with a half life of about 10 minutes
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
